As atmospheric CO2 increases, the ocean will become more acidic. The oceans are known to absorb about 1/3 of the CO2 we put into the air. Current projections indicate the pH of the ocean will drop by 1.4 over the next 300 years. Now keep in mind this is on a log-scale so the current ph of the ocean is between 8-8.3, which corresponds to 1/10 concentration of hydrogen ions. A drop of 1.4 puts the pH at 6.6-6.9 closer to hydrogen ion concentration of 10. This is between a 10x-100x increase in acidity.
When CO2 dissolves and reacts with water it can from three chemical species: 1) dissolved carbon dioxide, carbonic acid (H2CO3), bicarbonate (HCO3-), and carbonate (CO32-). The ratio depends on temperature and alkalinity. The short is that increasing CO2 results in increased carbonic acid causing a reduction of carbonate ions necessary for corals and snails, among other invertebrates.
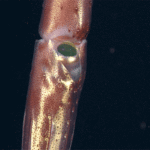
A recent Brevia in Science indicates that skeleton-producing corals can actually survive these acidic conditions maintaining basic life functions including reproduction. In the experiments after one month total skeleton dissolution occurred. The polyps themselves however elongated, dissociated from the colony, and affixed to hard substrate. Indeed, polyp biomass increased 3x that of polyps associated with calcified colonies. After 1 year, the solitary polyps were transferred back to normal conditions where the calcified and reformed colonies. Compared to organisms that survived mass extinction events by seeking geographic localities with favorable conditions, geographical refugia, the authors suggest that corals may use physiological refugia, switching between hard and soft forms, to survive stressful conditions.